Flexible containment solutions
Choose the right level of protection for your process—from basic dust control to advanced containment systems—ensuring safety, compliance, and smooth production.
Advancing pharma manufacturing with Gericke solutions
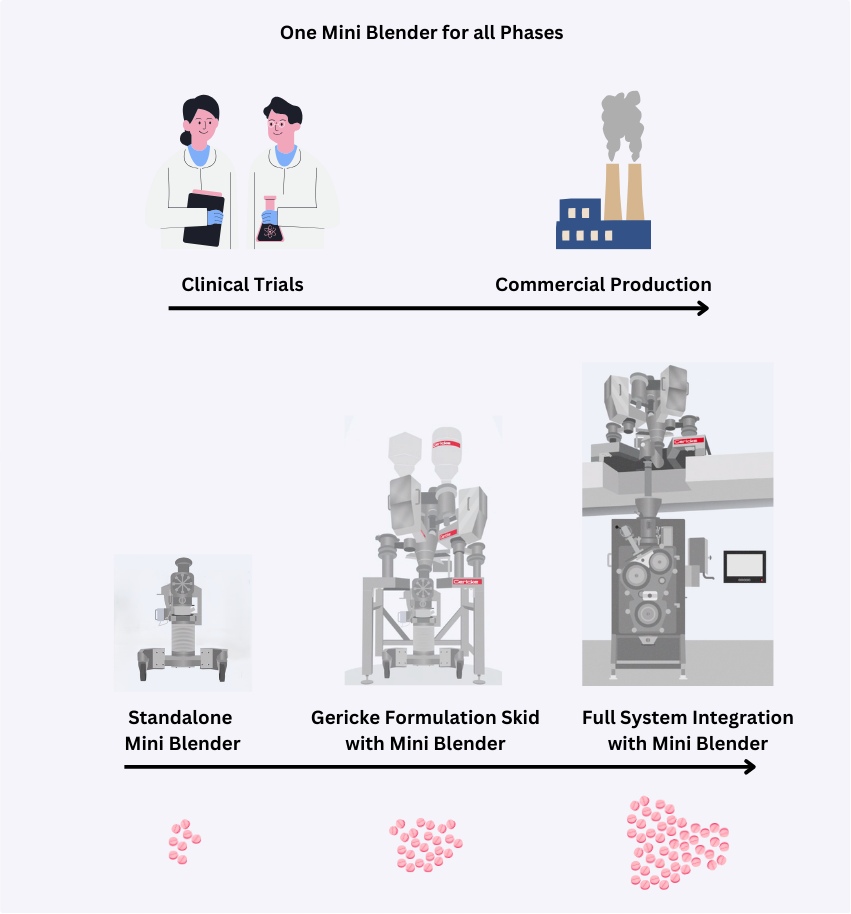
For the innovator market who has the need for a simple, fast and safe drug formulation, Gericke provides barrier-free continuous manufacturing equipment, in form of a single process solution from early pre-clinical trials to commercial production.
Get in touch!
Choose the right level of protection for your process—from basic dust control to advanced containment systems—ensuring safety, compliance, and smooth production.
Designed for simplicity, the system assembles and disassembles quickly with precise, repeatable alignment—reducing setup time and ensuring consistent performance every run.
Carry your formulation from clinical through commercial production on the same line and blender — avoiding the usual risks, delays and re‑validation that come with traditional scale‑up.
No need to re-engineer or re-qualify new equipment — simply run longer or in parallel, enabling rapid scale‑out, faster supply ramp-up and quicker time‑to‑patient with minimal disruption.

ADVANCED PHARMA
Mini blends, high containment, and full flexibility — this is continuous manufacturing for innovators. Engineered for complex formulations and regulatory confidence, our newest GFS is engineered with advanced containment and hygienic design. One platform and single process solution – from development to commercial launch.
SMALL BATCHES, BIG FLEXIBILITY
Our new GFS with integrated mini blender gives innovators the power to run small-scale production with full containment and GMP-grade hygiene. It’s the perfect bridge between R&D and production — enabling fast formulation iterations, reduced material use, and seamless scale-up when you're ready to go big.

FIRST STEP TO CONTINUOUS MANUFACTURING
Begin with trials at our test center or a Mini Blender for early adoption and proof-of-concept. Later, integrate it into a full continuous manufacturing system—using one validated process to reduce risk, speed scale-up, and accelerate time-to-patient.
Connect with us to find the right approach for you.
Connect with us to find the right approach for you.
The people make the difference! We are proud that we can rely on a truly global network of experienced professionals. Every day they show with their dedication that they are proud to be a part of the Gericke family and continue the success story of our company!