Science-driven partnerships
Our strong ties with leading scientific institutions mean your project benefits from cutting-edge research, real-world validation, and co-developed innovation

Benefit from years of powder handling expertise to enhance your continuous OSD production. Gericke experts support you through every project phase, from selecting a continuous production system to seamless integration, ensuring maximum efficiency.
Get in touch!
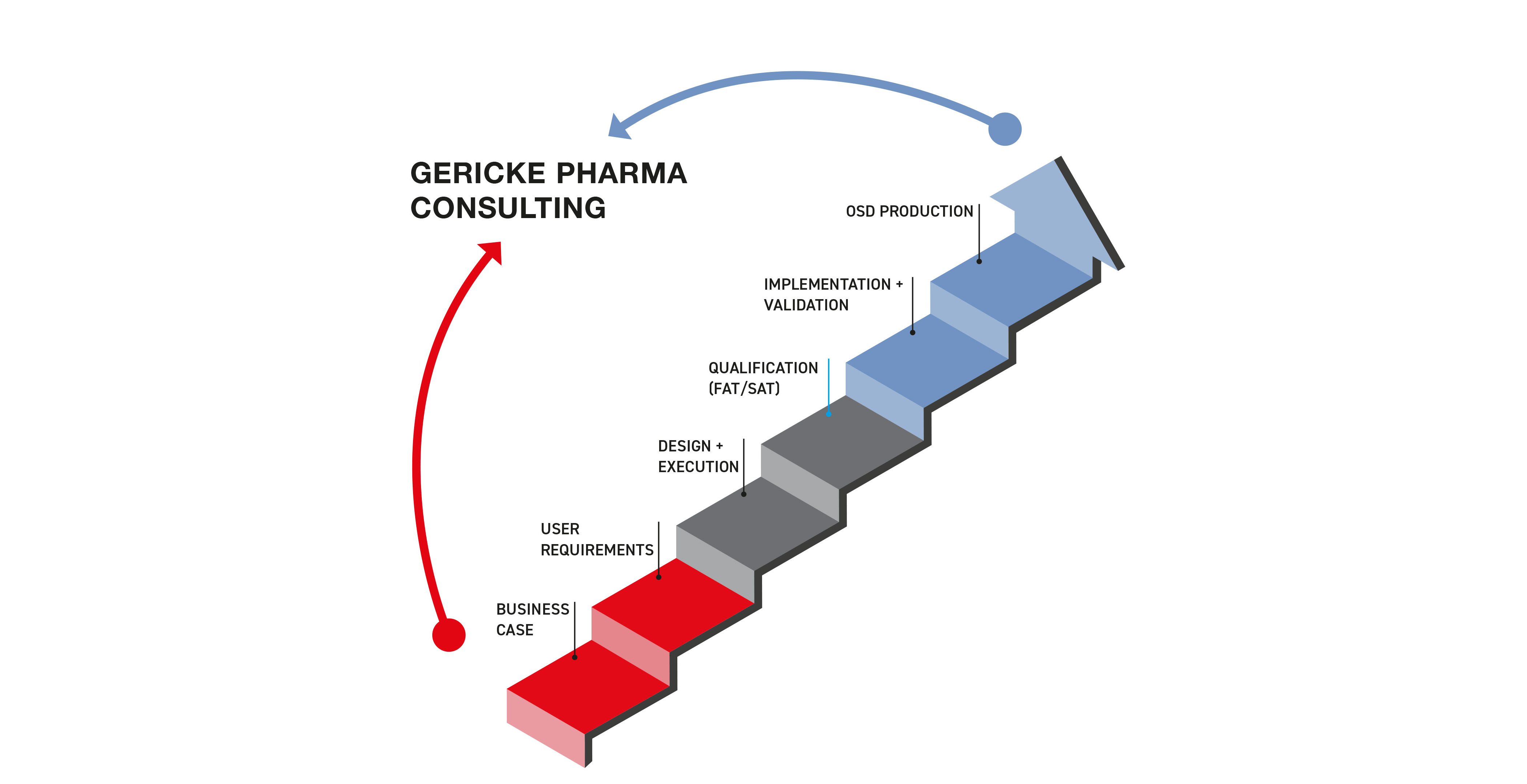
Pharma Consulting Services
We co-create smarter manufacturing.
From strategy to scale-up, we solve real-world challenges with data, process expertise, and resilient solutions.
Explore our consulting packages or start the conversation.
Make the case, confidently.
We help you quantify the benefits of switching to continuous manufacturing — from cost savings to process efficiency — so you can build internal alignment and secure buy-in.
Define what matters.
Together, we shape your process goals, product specs, and performance needs — laying the foundation for a solution that fits.
Design it right, build it smart.
We tailor your system to your space and process — ensuring feasibility, smooth execution, and future scalability.
Test before you trust.
We run full-scale FATs and SATs to validate your system — with real equipment, real data, and zero surprises.
From plan to production.
We guide installation, integration, and validation — making sure your system performs as promised.
Launch with confidence.
With optimized formulation and validated systems, you're ready to run continuous manufacturing at full scale.
Our strong ties with leading scientific institutions mean your project benefits from cutting-edge research, real-world validation, and co-developed innovation
From strategy to execution, we’re hands-on partners — not just advisors — guiding you through every step of the journey.
Our team blends process, automation, digitalization, and quality - so your solution works across every layer of your operation.
We simplify compliance for continuous manufacturing—bridging regulatory gaps and easing validation for a smooth transition.
VALIDATED SYSTEMS
We don’t just test parts — we test the whole thing.
Our FATs simulate your full pharma process, end-to-end, with real equipment and real data. We assemble the full system, run it like it’s already in production, and make sure it performs exactly as promised — before it ever leaves our floor.
Watch the system come together, run like clockwork, and roll out the door — ready for pharma.
confidence in seamless motion
Gericke gives pharmaceutical manufacturers the confidence to move into Continuous Manufacturing with precision‑engineered feeding, blending, and material‑handling systems that deliver stable, repeatable performance from development to commercial scale. Through our collaboration with DFE Pharma — and the integration of our technology into their new C2F (Closer to the Formulator) site in Hyderabad — customers can now explore, test, and validate continuous processes in a fully equipped, representative environment without upfront investment or operational risk.

A complementary combination that builds a strong foundation for reliable CM evaluation, smoother decision‑making, and confident scale‑up.
Discover how Gericke and DFE Pharma can streamline your path to CM: let’s talk!
Request a technical discussion: pharma.ch@gerickegroup.com
Reliable CM starts with excipients that flow consistently. When paired with precise feeding and blending technology, their optimized properties support smoother processing, reduced formulation complexity, and more stable, high quality production.
High precision feeding and blending systems enable rapid CM feasibility work, letting teams explore the technology without upfront investment or operational risk. Because the equipment used in trials mirrors real production scale setups, the learnings and process data transfer seamlessly — giving customers confidence that what works in evaluation will work in full implementation.
Years of combined process, formulation, and continuous manufacturing expertise help guide teams with clarity. With deep understanding of material behavior, equipment performance, and CM requirements, customers gain shorter learning curves, clearer direction, and confidence that both the technical decisions and regulatory pathways are grounded in proven experience.

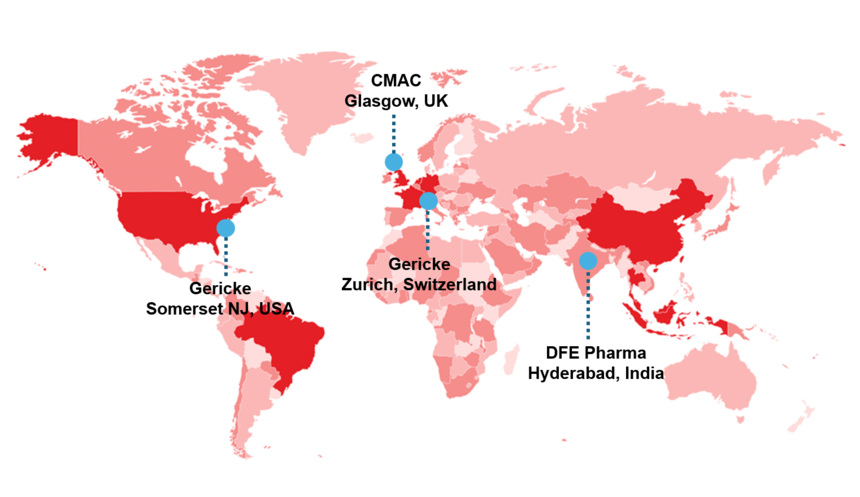
pharma test facilities
Step into continuous manufacturing at one of our globally distributed test centers. Together with our partners, we offer the ideal environment for innovation — combining state-of-the-art equipment with their formulation development expertise. Whether you’re running full-scale trials or exploring new formulations, you’ll have access to real machines, real data, and collaborative support from Zurich to Hyderabad.
our partners
Our continuous manufacturing expertise is backed by scientific partnerships and peer-reviewed research. Explore published papers that demonstrate proven results, innovative process design, and real-world applications—helping pharma manufacturers transition to smarter, more efficient production:

Continuous manufacturing offers faster production, improved product consistency, and real-time process control. Unlike batch processing, it enables a single validated process that can scale from development to commercial production—reducing time-to-market and regulatory complexity
Yes. Gericke’s precision feeding and blending technologies are designed for high containment and accuracy, making them ideal for high-potency APIs and complex formulations. Our systems ensure consistent quality while maintaining operator safety and regulatory compliance
Gericke’s modular systems adapt to a wide range of formulations and throughput needs. With options like the Mini Batch/Blend setup, customers can start small and scale up using the same validated process—minimizing risk and maximizing flexibility
We guide you from concept to implementation, offering process expertise, equipment integration, and validation support. Our low-barrier entry solutions like Mini Batch/Blend allow for a smooth transition, enabling proof-of-concept without disrupting existing operations.
Absolutely. All Gericke systems are designed to meet global GMP requirements and support compliance with FDA and EMA guidelines. Our process transparency and control features simplify documentation and validation for regulatory approval
Contact Information Continuous Manufacturing

Head of Continuous Manufacturing (OSD)

Consulting Lead Continuous Manufacturing (OSD)

Process Engineer Continuous Manufacturing (OSD)

Product Manager Continuous Manufacturing (OSD)

The people make the difference! We are proud that we can rely on a truly global network of experienced professionals. Every day they show with their dedication that they are proud to be a part of the Gericke family and continue the success story of our company!